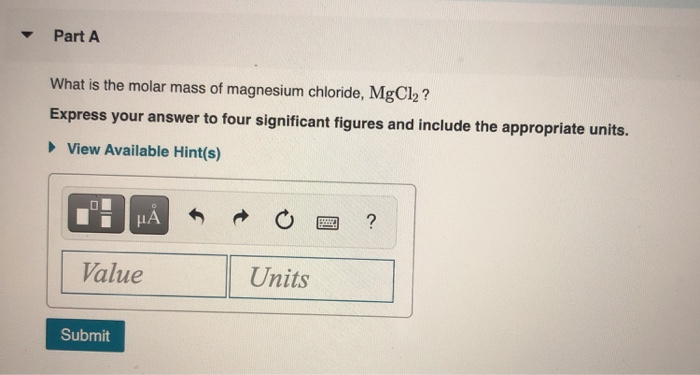
In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol.  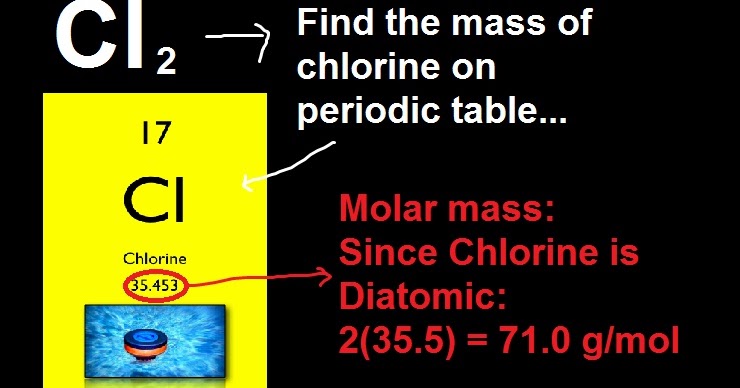
This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. = (14.01 amu * 2) + (1.008 amu *4) + (16.Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance. The atomic mass of an element (in atomic mass units – amu) is equal to its “molar mass” Molar masses: Different atomic masses lead to differences in molar masses. Basis of Calculationįormula mass: Different chemical elements in the chemical compounds give different formula masses. Molar mass: Molar mass is calculated for chemical substances that contain many elementary entities such as chemical elements, ionic and covalent chemical compounds. It is calculated using the empirical formula. Useįormula mass: Formula mass is calculated for chemical compounds. Molar Mass: Molar mass is the mass in grams of 1 mol of substance (The number of particles in a mole is equal to 6.022 * 10 23). What is the difference between Formula Mass and Molar Mass? Definition of Formula Mass and Molar Massįormula Mass: The formula mass (formula weight) of a molecule is the sum of the atomic weights of the atoms in its empirical formula. What is the formula mass of NaCl (Atomic mass of Na = 22.9898 amu, Atomic mass of Cl = 35.4527 amu )? The units of formula mass “ atomic mass unit” (amu). The formula mass of a molecule or a compound is the sum of the atomic weights of the atoms in the empirical formula. The key difference between formula mass and molar mass is that, the formula mass of a molecule or a compound is the sum of the atomic weights of the atoms in its empirical formula while molar mass is the mass in grams of 1 mol of substance. It is the sum of the atomic mass values of individual components in the empirical formula (H 2O-18.00 g mol -1). Formula mass is calculated considering the empirical formula of a compound. Similarly, this leads to having various unique molar mass values for chemical compounds ( NaCl–58.4426 g mol -1). Different chemical elements have different molar mass values (C -12.01 g mol -1, Mg-24.3050 g mol -1) since they have a different number of positions in the nucleus. Therefore, the mass of 1mol (mass of 6.02 * 10 23 particles in grams) is used as the unit in quantitative analysis. Since atoms, molecules, and unit cells are extremely small particles the mass of one particle is negligibly small. Both of these parameters, the formula mass and molar mass, are related to the weight of chemical elements (atoms, molecules, unit cells). Key Difference – Formula Mass vs Molar Massįormula mass and molar mass are two physical properties of molecules that show some difference between them.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
